| Great Lakes Ecosystems | ||

|
|
|||||||||
|
1994
Proceedings
|
|
PHENOLOGICAL AND PHYSIOLOGICAL RESPONSE OF SHOOTING
STAR (DODECATHEON MEADIA L.) IN PRAIRIE AND SAVANNA HABITATS Dan Gustafson and Roger C. Anderson |
 |
There has been considerable interest in recent years in defining characteristic species of savannas. Some investigators have proposed that savannas are composed of species assemblages which are relatively distinct from woodlands and prairie. There are, however, many species which commonly occur in prairies and savannas or woodlands and savannas (Curtis 1959). Because these habitats provide different environmental conditions for plants, it might be expected that species occurring across the prairie-savanna-woodland gradient might be composed of individuals adapted to portions of this gradient.
The purpose of this study is to determine if there are physiological and phenological differences between plants of the same species occupying prairie and savanna habitats, using shooting star (Dodecatheon meadia L.) as the test species. The savanna with its associated tree canopy presents a habitat with a reduced period of time to which plants are exposed to full solar irradiation. This could have an effect on the phenological and physiological adaptations of the savanna biotype. For example, the savanna biotype may exhibit a faster rate of phenological development than the prairie biotype, allowing the savanna biotype to complete its life cycle before the savanna tree canopy closes. The savanna biotype may be better adapted to carry out photosyn-thesis under conditions of reduced solar irradiation than the prairie biotype. These differences in biotypes should be reflected in the chlorophyll content of the leaves, leaf structure, and the photosynthetic rates of the prairie and savanna biotypes under various solar radiation intensities (Boardman 1977). In this study, we examined phenological development, leaf characteristics and photosynthetic rates, under varied irradiance levels, of savanna and prairie populations of shooting star.
The prairie site is the 2.1 ha Weston Cemetery Prairie located in McLean County, Illinois. With the exception of grave sites, the prairie has experienced little physical disturbance. The vegetation is thought to be representative of the mesic tallgrass prairies that once occupied much of central Illinois prior to European settlement (Johnson and Anderson 1986). This small prairie remnant is surrounded on three sides by agricultural crops; to the north there is a railroad right-of-way which is bordered by another agricultural field. The management program includes a biennial burning regime as a method of maintaining the prairie species composition with one-half of the prairie being burned each year. The west one-half of the prairie was burned on 7 April 1993; the burned and unburned sections of the prairie were treated as separate study plots.
The savanna study site is located on the ParkLands Foundation Merwin Nature Preserve 7.0 km west of Lexington, Illinois. This area is currently being restored to pre-European settlement savanna condition. The canopy currently consists of oak and hickory trees with the surrounding areas consisting of secondary growth forest. Since 1967, cutting and grazing were removed from the site. Elimination of these disturbance regimes and fire protection led to the invasion of woody species, which converted the site to a nearly closed canopy forest. The savanna restoration began in 1989 and consisted of selective removal of tree species and annual prescribed burns to open the canopy and encourage the growth of the remnant savanna species.
Ten shooting star plants were located and marked at the prairie and savanna sites on10 April 1993. To monitor individual plant growth, leaf surface area was estimated by measuring the maximum leaf length and width of each leaf in the basal rosette, at weekly intervals. To establish a relationship between leaf length, width and area for each sampling date, 3-8 leaf tracings were made at each site from leaves depicting the range in leaf size on that date. A computer aided image processing system (Image-pro Plus) was used to determine the surface area of each leaf tracing. Regression analysis was performed to establish the relationship between the product of leaf length, width, and surface area. Senescence data were obtained from the same plants by estimating the percent green leaf remaining for each plant at approximately weekly intervals. For each plant, leaf area measurements made at each sampling date were expressed as a percent of the maximum leaf area. For each measurement date, a life cycle index was calculated by adding percent full leaf expansion and percent senescence for each plant and dividing by two. The index ranges from zero to 100%; it is 50% when full leaf expansion occurs and 100% when a plant has undergone complete senescence. These data were then regressed over time for each site. Student's t-tests were used to test for significant (p<0.05) differences among the slopes of the three lines (Steel and Torrie 1960).
The chlorophyll extractions and measurements were made following the procedures of Vernon (1960). These data were analyzed by an analysis of variance (ANOVA) using the SPSS-PC statistical package (SPSS/PC+ 1990).
When plants achieved full leaf expansion, rates of photosynthesis from individual leaves were measured using the LI-COR 6000 gas analyzer. Plants were exposed to varied levels of solar radiation consisting of full solar radiation and two levels of shade (0.50 and 0.25 of full solar irradiation) achieved by means of one and two layers of shade cloth, respectively. Photosynthetic measurements were conducted during the hours of maximum solar radiation (11:00 am to 1:00 pm) on days with no cloud cover. Photosynthetic rates were calculated using LI-COR 6200 linear regression equations, which increase the reliability of the LI-COR 6000 estimates of photosynthesis (McDermitt 1987). The photosynthetic data included carbon dioxide concentration, relative humidity, leaf and chamber temperatures, photosynthetically active radiation. Photosynthetic rates were analyzed using a multivariant statistical analysis procedure (MANOVA) with repeated measures (SPSS/PC+ 1990).
There was a significant positive correlation between the product of leaf length and width and leaf area (r squared = 0.994, 0.989, and 0.975 for the ParkLands, Weston burned, and Weston unburned sites, respectively (p<0.05 in all cases)). Slopes of the life cycle regression analyses were all significantly different. The ParkLands savanna population had the fastest rate of phenological development. This shortened life cycle allowed the savanna biotype to complete most its life cycle before the savanna tree canopy closed. Weston burned population had a faster rate of phenological development than the unburned population. An increase in the rate of development and productivity following a burning treatment in a tallgrass prairie have been previously documented (Old 1969; Knapp 1984).
The concentration of the total chlorophyll, chlorophyll A and chlorophyll B were not significantly different among sites. However, chlorophyll A/B ratios were significantly different among all three sites (Table 1). A lower A/B ratio is indicative of plants that have developed in a shaded environment (Knapp and Gilliam 1985). Consistent with this expectation, ParkLands savanna plants the lowest A/B ratio. Plants of Weston unburned site had the second lowest A/B ratio, and their initial development occurred in a shaded environment. Early season growth of vegetation in unburned, ungrazed tallgrass prairie occurs in a very low-irradiance environment due to the accumulation of a large amount of standing dead biomass (Old 1969; Knapp 1984). Once the plant has grown above the litter level, the leaves are exposed to levels of full photosynthetically active radiation. However, prairie plants developing in an initial shaded environment may develop several morphological and physiological characteristics of shade leaf, including modification of the chlorophyll concentrations (Knapp and Seastedt 1986). Plants from Weston burned site possessed the highest A/B ratio which is consistent with plants that develop under full light intensities.
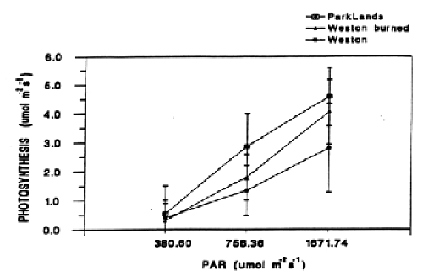
For each light regime, there are no significant differences in the amount of photosynthetically active radiation, temperature, relatively humidity, and the concentration of carbon dioxide under which savanna and prairie plants were measured. ParkLands savanna demonstrated a higher rate of photo-synthesis than that of prairie sites, but these differences were not significant (Fig. 1).
In summary, the savanna biotype had a somewhat shortened life cycle compared to the prairie biotype. The savanna plants possessed the lowest A/B ratio followed by the unburned prairie plants, which developed in the initial shaded environment, and burned prairie plants, which had the highest A/B ratio. The photosynthetic analysis determined no significant difference in the rates of photosynthesis between the prairie and savanna biotypes.
I thank Dr. V. Borowicz and Dr. R. Preston for the use of equipment and the Beta Lambda chapter of Phi Sigma for financial support.
Boardman, N. K. 1977. Comparative photosynthesis of sun and shade plants. Annual Review of Plant Physiology 28: 355-377.
Curtis, J. T. 1959. The Vegetation of Wisconsin: An Ordination of Plant Communities. University of Wisconsin Press, Madison, WI. 657 p.
Johnson, R. G. and R. C. Anderson. 1986. The seedbank of a tallgrass prairie in Illinois. American Midland Naturalist 115: 123-130.
Knapp, A. K. 1984. Post-burn differences in solar radiation, leaf temperature and water stress influencing production in a lowland tallgrass prairie. American Journal of Botany 71: 220-227.
Knapp, A. K. and F. S. Gilliam. 1985. Response of Andropogon gerardii (Poacea) to fire-induced high vs. low irradiance environments in the tallgrass prairie: leaf structure and photosynthetic pigments. American Journal of Botany 72: 1668-1671.
Knapp, A. K. and T. R. Seastedt. 1986. Detritus accumulation limits productivity of tallgrass prairie: the effects of its plant litter on ecosystem function make the tallgrass prairie unique among North America biomes. Bioscience 36: 662-668.
McDermitt, D. K. 1987. Photosynthetic measurement systems: performance comparison of the LI-6200 to the LI-6000. Application notes 6200-1. LI-COR Inc., Lincoln, Nebraska.
Old, S. M. 1969. Microclimate, fire, and plant production in an Illinois prairie. Ecological Monographs 39: 355-384.
SPSS/PC+. 1990. SPSS/PC+ 4.0 Base Manual. SPSS Inc., Chicago, Illinois.
Steel, R. G. D. and J. H. Torrie. 1960. Principles and Procedures of Statistics. McGraw-Hill Book Company, Inc., NewYork, NY.
Vernon, L. P. 1960. Spectrophotometric determination of chlorophylls and pheophytins in plant extracts. Analytical Chemistry 32: 1144-1150.
Table 1. Leaf chlorophyll concentration of Dodecatheon meadia (mg / g fresh weight). Within a row, means with the same letter are not significantly (p<0.05) different.
| Character | Parklands Savanna |
Weston Prairie Burned |
Weston Prairie Unburned |
| Total Chlorophyll (mg/g) |
0.844 +/- 0.060 a | 0.863 +/- 0.043 a | 0.801 +/- 0.061 a |
| Chlorophyll A (mg/g) |
0.591 +/- 0.042 a | 0.625 +/- 0.028 a | 0.570 +/- 0.047 a |
| Chlorophyll B (mg/g) |
0.253 +/- 0.018 a | 0.238 +/- 0.016 a | 0.230 +/- 0.014 a |
| A:B ratio (ratio) |
2.332 +/- 0.021 a | 2.640 +/- 0.075 b | 2.464 +/- 0.060 c |
Figure 1. Photoshythesis vs. photosynthetically active radiation (PAR). Means plus or minus one standard error for three levels of PAR.