PRELIMINARY FINDINGS OF THE ECOLOGICAL COMMITTEE ON FIFRA RISK ASSESSMENT METHODS (ECOFRAM): VI. AQUATIC EFFECTS ANALYSIS
ECOFRAM Aquatic Effects Subcommittee: Jeff Giddings, chair (Springborn), Larry Barnthouse (LWB Environmental), David Farrar (EPA), Tilghman Hall (Bayer), Mike McKee (Monsanto), Mike Newman (VIMS), Kevin Reinert (Rohm & Haas), Bob Sebastien (Environment Canada), Keith Solomon (U. Guelph), Ann Stavola (EPA), Les Touart (EPA), Randy Wentsel (EPA)
On this Page
- Abstract
- Factors Contributing to Uncertainty in Analysis of Ecological Effects of Pesticides
- Tiers 1 and 2
- TIER 3: A Toolbox of Options for Reducing Uncertainty About Ecological Effects
- TIER 4: Focused Investigations to Address Specific Risk Assessment Questions
Abstract
The Aquatic Effects Subcommittee of ECOFRAM has explored a variety of tools and approaches for reducing uncertainty in the analysis of pesticide effects in aquatic risk assessments.
Generic life-table methods and age/stage models can be used to extrapolate from individual-based measurement endpoints such as survival, growth, and reproduction to population-based assessment endpoints such as recovery time and likelihood of local extinction; more complex, species-specific population models can be applied at higher tiers of a risk assessment.
Species sensitivity distributions can be analyzed to estimate risk to untested species and effects on community taxonomic richness.
Several approaches can be used to evaluate the effects of time-varying exposure (which is the typical case for many pesticides):
- time-to-event analysis of conventional toxicity data,
- extended laboratory toxicity tests using pulsed concentrations,
- uptakedepuration models coupled with dose-response models based on critical body burden, and
- population models to predict effects of repeated exposures.
Other tools that can be used to address specific issues at higher tiers of a risk assessment include
- sediment toxicity tests,
- behavioral toxicity tests, and
- model ecosystems (microcosms and mesocosms).
ECOFRAM is now working to integrate these tools into a logical sequential risk assessment process (see Poster VII).
Factors Contributing to Uncertainty in Analysis of Ecological Effects of Pesticides
Factors influencing individual sensitivity
- Age, size, life stage
- External conditions (temperature, hardness, etc.)
- Individual condition (nutrition, health)
- Genetics
- Endpoint selection and calculation (LC50, EC5, NOEC, biomarkers)
Time-varying exposures
- Toxic effects on individuals (latency, reciprocity, reversibility)
- Population effects
Effects of contaminated sediment on benthic organisms
Behavioral effects
Interspecific differences in sensitivity (extrapolation to untested species)
Extrapolation from individual effects to population consequences
Extrapolation from individual/population to community/ecosystem
- Recovery (individual, population)
- Recolonization (refugia, immigration)
- Functional replacement
- Population interactions (food web, keystone species, habitat)
Multiple stressors
- Toxic effects on individuals
- Population effects
- Community/ecosystem effects
Tiers 1 and 2
Standard Toxicity Tests
Acute toxicity to freshwater algae (1 or more species-new 40 CFR 158 data requirements may call for 4 species of algae plus duckweed for all pesticides, but ECOFRAM considers one species sufficient for insecticides). Endpoint: 96-hour EC50 (14-day EC50 for duckweed).
Acute toxicity to Daphnia. Endpoint: 48-hour EC50.
Acute toxicity to freshwater fish (warm water, cold water). Endpoint: 96-hour LC50.
Chronic toxicity to Daphnia (life cycle). Endpoint: 21-day EC10. a
Chronic toxicity to freshwater fish (early life-stage). Endpoint: 35-day or 90-day EC10.a
If there is potential for marine exposure:
Acute toxicity to marine algae. Endpoint: 96-hour EC50.
Acute toxicity to marine arthropod (e.g., mysid). Endpoint: 96-hour EC50.
Acute toxicity to marine mollusc. Endpoint: 48-hour or 96-hour EC50.
Acute toxicity to marine fish. Endpoint: 96-hour LC50.
Chronic toxicity to mysid (life cycle). Endpoint: 28-day EC10.a
Chronic toxicity to marine fish (early life-stage). Endpoint: 35-day or 90-day EC10.a
a The No Observed Effect Concentration (NOEC) can be used if regression results in poor EC10 estimate, provided the study is otherwise acceptable.
Notes
ECOFRAM recommends including invertebrate life cycle studies and fish early life-stage studies in Tier 1, even though they are only conditionally required under 40 CFR 158. These studies are nearly always conducted anyway. Including them in Tier 1 allows for a more thorough assessment and reduces the time needed to reach a registration decision.
The precision and accuracy of acute and chronic mortality endpoints can be improved using Time-to-Event (TTE) analysis (see sidebar).
In Tier 2, the entire exposure-response relationship (not just a point estimate like the LC50) is used in the risk characterization.
Population analysis (e.g., life table analysis or age/stage population models) for generic life history types can provide perspective on the population-level implications of individually-based toxicity endpoints (see example at right).
Time-to-Event Analysis
Objective: Assess effects as a function of exposure duration as well as concentration
Input: Observed effect over a range of exposure times
Output: Equation relating magnitude of effect to concentration and exposure duration. For example:
TTE = ea elog(C) eε
where
TTE = time to event,
C = concentration,
a = constant, and
ε is an error term.
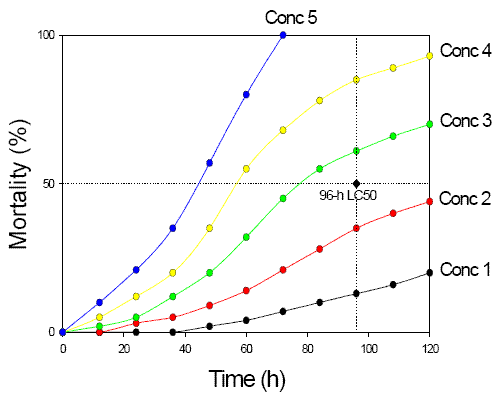
Time-to-Event Analysis Graph
Advantages
- Allows estimation of effects to match time scale of exposure
- Compatible with standard acute testing procedures
- Enhanced statistical power
Limitation
- Doesn't account for latent effects
Generic Population Analysis: Example
Analysis based on a simple model of exponential (density independent) population growth:
Nt = N0 ert
where
Nt = number of organisms present at time t,
N0 = number of organisms present at time zero, and
r = intrinsic rate of increase (per unit time).The time (tr) required to replace a fraction (X) of the population (lost, for example, through mortality caused by pesticide exposure) is calculated as
tr = ln (1/(1-X))/r.
The figure below shows the time to recovery for hypothetical daphnid populations with r = 0.15 or r = 0.3, as a function of percent mortality.
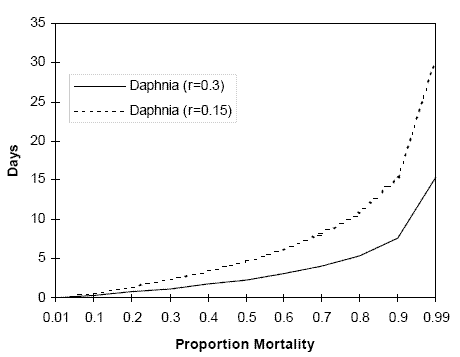
Generic Population Analysis Graph
TIER 3: A Toolbox of Options for Reducing Uncertainty About Ecological Effects
Sensitivity Distributions
Measure acute toxicity to additional species-fish, invertebrates, or plants, as indicated in Tiers 1 & 2. The number of species tested will vary depending on the needs of the risk assessment. A total of 8 toxicity values is usually enough to describe the sensitivity distribution accurately.
Developing toxicity data on additional species reduces the uncertainty of extrapolating from tested species to other species.
Analysis of species sensitivity distributions allows estimation of the fraction of species (at a site, in a region, or globally) that might be affected at different exposure levels.
Acute toxicity data on a broader range of species can be useful for site- specific ecological assessment.
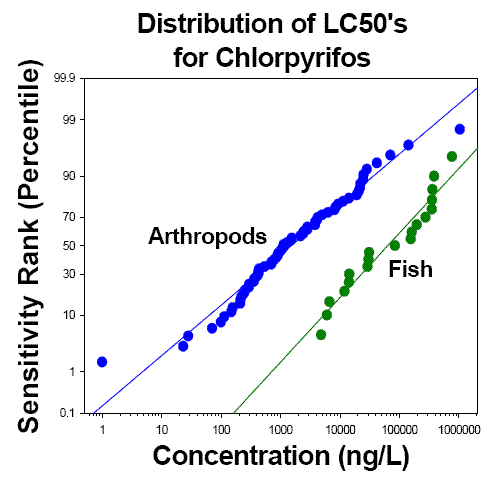
Distribution of LC50's for Chlorpyrifos
Time-Varying Exposure
Standard acute and chronic toxicity tests are designed to measure the effects of exposure to constant chemical concentrations. However, pesticide concentrations under field conditions typically vary over time (see example below). Pesticide concentrations may decline after application due to chemical degradation, biodegradation, partitioning to sediment or organic matter, and hydrologic dilution and dissipation. If exposure assessment in Tiers 1 and 2 indicates that these factors are likely to be important, the effects of time-varying exposure should be investigated.
- Shorter exposure may have less effect than exposure of standard duration.
- Pulses could select for less sensitive individuals, so later pulses would have less effect.
- Pulses could weaken survivors (cumulative damage), so later pulses would have more effect.
- Pulses could strengthen survivors (acclimation), so later pulses would have less effect.
- Effects could occur after exposure ends (latency).
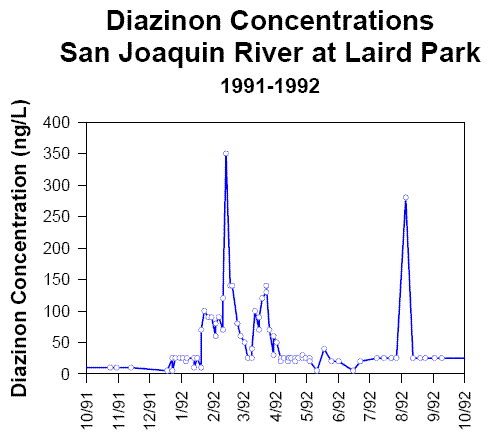
Diazinon Concentrations
San Joaquin River at Laird Park
1991-1992
Strategies for assessing effects of time-varying exposures
- Laboratory toxicity tests with pulsed or dissipating exposure regimes.
- Bioaccumulation models (based on uptake and depuration kinetics) coupled with information on internal dose-response relationship (critical body burden).
- Advanced (data-intensive) toxicokinetic models such as FGETS and PULSETOX (more appropriate in Tier 4-see below).
- Population models simulating repeated impacts separated by intervals of recovery.
Chronic Toxicity Tests
Development of chronic toxicity data for additional species is recommended if
- chronic risk is demonstrated in lower tiers
- prolonged or repeated exposure is expected
- the compound has a potential for bioconcentration (high Kow or measured BCF)
- the mode of action or other data suggest that chronic effects may occur
Testing may include fish full life-cycle tests or additional chronic studies with invertebrates, depending on sensitivity patterns revealed in Tiers 1 and 2. The appropriate exposure regime will be determined based on information generated in the exposure analysis.
Sediment Toxicity Tests
The decision to focus on sediment toxicity is based on acute and chronic risk characterization using pore-water concentrations (as determined by an exposure model) and invertebrate toxicity tests in Tiers 1 and 2. Pore-water predictions take into account the factors influencing pesticide accumulation in sediment, such as Koc and sediment organic content.
If sediment toxicity is found to differ substantially from predictions based on pore-water concentrations, unknown factors may be influencing bioavailability; tests with additional sediment types may be needed.
TIER 4: Focused Investigations to Address Specific Risk Assessment Questions
Options Include
Pharmacokinetic Models
Physiologically-based pharmacokinetic (PBPK) or toxicokinetic (PBTK) models describe absorption, distribution, metabolism, and excretion. Their principal application is prediction of dose to a target tissue, or body burden for parent chemical or transformation product over time. Coupling these models with critical body residues (CBRs) permits estimation of when a chronic or acute toxic response will occur under various exposure scenarios (including time-varying or repeated exposure-see Tier 3). These models can be very data intensive.
Population and Ecosystem Models
Population models used in Tier 4 would be highly species- or environment- specific. Individual-based models, metapopulation models, or spatially explicit models could be used. The models would be specifically designed to address the uncertainties (such as regional variability, effects on endangered species, indirect effects, or unusual modes of action) that led to the Tier 4 assessment.
Models of ecosystem structure and function may have value on a case-by-case basis.
Microcosms and Mesocosms
Microcosms and mesocosms have several uses in the highest tier of a risk assessment
- Incorporate quasi-realistic exposure including pesticide partitioning and dissipation;
- Measure responses of many taxa;
- Observe population, community, and ecosystem responses;
- Observe ecological recovery.
These studies can be expensive to conduct, produce complex results, and can have high variability (like nature). They are useful to address "what if?" and "so what?" questions.
Behavioral Tests
Behavior is a manifestation of physiological and biochemical processes, and as such can act as a sensitive indicator of exposure to a toxicant. Behavioral tests could be designed to answer an ecologically relevant question with an interpretable response. For example, toxicant avoidance may decrease exposure, but does this avoidance have implications due to the displacement of the organism from beneficial habitat?
![[logo] US EPA](../gif/logo_epaseal.gif)