Modeling Subsurface Petroleum Hydrocarbon Transport
| Module Home Objectives Table of Contents Previous < Next > |
| 2 of 22 |
|
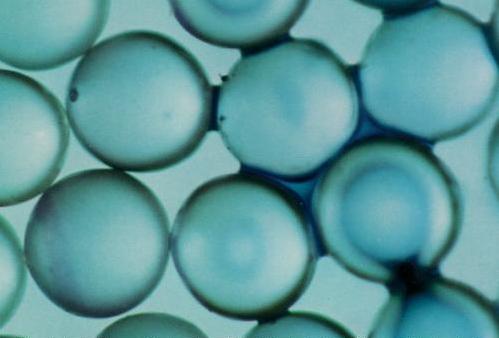
The picture to the left shows water and air in a glass bead pack. The diameter of the glass beads ranged from 0.85 to 1.23 mm. The water, which was dyed blue, can be seen in the small pores and contacts between the beads. This distribution of water and air, and the concave interfaces show that the water is being pulled toward the solid surfaces. This indicates that the water is more strongly attracted to the solid surfaces than is the air. |
|
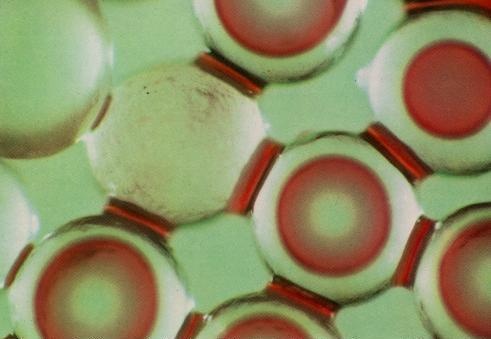
Now there is NAPL (perchloroethene) and air in a similar bead pack. The perchloroethene has been dyed red. No water is present in this picture. The NAPL phase is found in the small pores and contacts between the beads. Again the interfaces are pulled toward the solid surfaces, indicating that the NAPL is attracted preferentially toward the solids. When only NAPL and air are present, the NAPL phase behaves similarly to the water in the water and air system. |
|
In both cases (water/air and NAPL/air) the liquid phase is more strongly attracted to the solid surfaces than is the gaseous phase. Generally, this is because of the stronger forces between molecules of solids and liquids than solids and air. These isolated rings of liquid are called residual saturations, because when a fluid does not occupy a fully-connected path through the porous media it cannot flow. The term residual is used because the fluid is the residual amount remaining after flow has ceased. "Saturation" means the fraction of the pore space occupied by the fluid. A water saturation of 0.30 means that 30% of the pore space is occupied by water. |
|
|
Color photographs on this page reprinted with permission from Dense Chlorinated Solvents, Friedrich Schwille, Translated by James F. Pankow, 1988, Copyright CRC Press, Boca Raton, Florida c 1988 |
|
Home | Glossary | Notation | Links | References | Calculators |